
The name carbon tetrachloride implies one carbon atom and four chlorine atoms so the formula is CCl 4. How many chlorine atoms would be in a compound with the name tetrachloride? The compound Fe2 O3 contains five atoms in each molecule of the compound. If there are two of them that’s four total chlorine. Calcium chloride formula units each one has to chlorine. We have 12 chlorine atoms the next one if we have to. We recognize that tetra means four so one molecule contains four There three of them. How many chlorine atoms are in 4 calcium chloride formula units? 2 mole of oxygen atoms per mole of oxygen atoms = 2 x 6.02 x 10 23 oxygen atoms. Molar mass of M,COs - 0.01186 M 84.3 g/mol Since the compound. 1 mole of carbon atoms per mole of carbon dioxide = 6.02 x 10 23 carbon atoms. (d) (d) (a) Hints and Solutions ))))) Given percentage of chlorine in an hydrocarbon. How many moles of carbon atoms are in 1 mole of carbon dioxide?ġ mole carbon dioxide contains 6.02 x 10 23 molecules. This number is known as Avogadro’s number. The number of atoms of carbon-12 present in this one mole sample is 6.022 136 7 x 10 23. How much is a mole of carbon?Įxactly 12 grams of pure carbon-12 powder is known as one mole. In 1 mole chlorine there are 2 chlorine atoms. 
They form a molecule per mole in an element.How many atoms of chlorine are in 1 mole of chlorine gas? Here a molecular weight is the sum of the total mass in grams and atoms. The chapter 3 science class 9 notes pdf explains the concept of substance which has mass and occupies space. On the other hand, molar mass pertains to the weightage of one sample mole. The aggregate of all atomic masses contained in a molecule is held to be molecular mass. What is a Molecular Mass and Molar Mass in Class 9 Ch 3 Science Notes? The measurement of atomic mass is represented through the Atomic Mass Unit (AMU). It is an approximate equivalent to the number of neutrons and protons present. What are Atomic Mass and Atomic Mass Unit in Class 9 Science Notes Chapter 3? The total mass of a single atom amounts to Atomic Mass. In chemical combination, atoms of various elements undergo combination in order to form compounds. The mass spectrum of chlorine is good example of molecular element. In a particular element, all atoms are identical in both physical and chemical propertiesĪtoms of different elements vary in both physical and chemical properties There is no creation or destruction of atoms Very small particles called atoms constitute all matter What Does Daltonic Theory Define in Class 9 Chemistry Chapter 3 Notes? Class 9th science chapter 3 notes define Daltonic theory in details – 
There are specific symbols to represent the mass number and the atomic number of an element. Formula Mass (atomic mass unit, amu) The sum of the average atomic masses of all. The letter ‘Z’ represents the mass number of an element that is equal to the total number of neutrons and protons of the element. What is the molar mass o water oxyqen o chlorine 17 Fill in the chart. The atomic number ‘A’ of an element is the number of protons present in it. 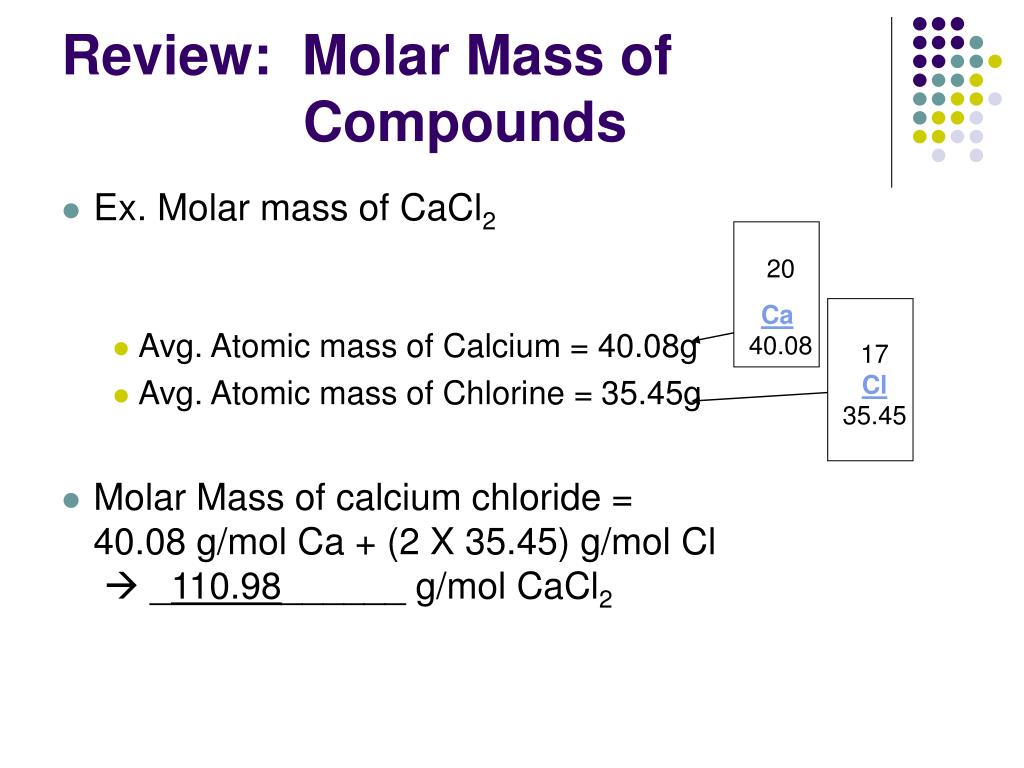
Law of Constant roportion: This law was expressed by another French chemist, Joseph Proust, as follows: "A chemical compound always comprises the same elements mixed in the same proportion by mass." Law of Multiple Proportions: As established by John Dalton, when two elements combine to form two or more compounds, the mass of the element that combines with the fixed mass of the other bears a simple whole-number ratio $\left( \text$ĬBSE Class 9 Science Notes Chapter 3 Atoms And Molecules What is an Atom in Chapter 3 Science Class 9 Notes? An atom is the smallest constituent of an element, which cannot be broken down further by any chemical reaction. Enter formulas with proper capitalization and unpack brackets. Table of Contents Law of Conservation of Mass: In a chemical action, the law of conservation of mass stipulates that mass can not be created or destroyed.According to this law, the overall mass of the products remains equal to the total mass of the reactants after any physical or chemical change. Molar Mass of Cl2 (Chlorine) (Our Other Math
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
